Introduction
As the target organization to consider, the American corporation Pfizer will be evaluated, and its target product is the COVID-19 vaccine. Determining the promotion context involves identifying its marketing goals, analyzing the industry segment, and highlighting the target markets. To assess the current operating conditions in its niche, Pfizer’s performance will be evaluated by utilizing a SWOT analysis and the analysis of competitors’ activities. Identifying relevant market factors is essential in determining the company’s current position and the success of its main product’s promotion.
Establishing the Context
Marketing Goals
One of the main goals the company has set is the massive and affordable distribution of the COVID-19 vaccine. In 2020, in accordance with the government program, the aim was to deliver 300 million doses of this vaccine to the market in 2021, which was decided to implement through expanded technological capacities and advanced logistics (Pfizer, n.d.). Another goal to achieve is to establish a partnership with BioNTech, Pfizer’s formal competitor. In early 2021, an initiative was announced to combine the efforts of the American and German corporations to ensure that as many vaccines as possible were and delivered to the target market (Pfizer, n.d.). Finally, continuous work to improve product quality is a valuable initiative to accomplish. Vaccine trials are ongoing, and the task of the company’s employees is to ensure the maximum safety and resistance of the product components to external conditions, thereby minimizing risks to the population (Pfizer, n.d.). These objectives are relevant solutions that reflect Pfizer’s commitment to the work performed.
Industry Segment
In accordance with the target activity, Pfizer’s industry segment can be analyzed based on the relevant characteristics. In terms of demographics, people from 50 to 64 are the main target audience, followed by 25 to 39 and 65 to 74, respectively (Centers for Disease Control and Prevention [CDC], 2021). Regarding gender composition, the female population who has received the vaccination slightly exceeds the male population (CDC, 2021). According to Sagonowsky (2021), by the end of May 2021, the corporation had expected to deliver 200 million doses, which covered its planned market size by two-thirds. By health status, adults and older people with chronic diseases are the target customers of Pfizer’s vaccine, and children constitute the smallest audience (CDC, 2021). According to the psychographic data, approximately the same ratio remains: adult and older people with chronic illnesses fear for their health (CDC, 2021). Children and young adults, in turn, make up a smaller percentage due to a more active lifestyle and less anxiety.
Pfizer’s target activities are government-controlled and accountable to the authorities. In addition, as regulatory influences, one should note state healthcare agencies and the Food and Drug Administration (FDA) (“Covid,” 2020). The customer base includes not only individual patients but also organizations. The largest number of customers in the first quarter of 2021 was from the financial services industry, followed by the property and casualty insurance industry and accident and health insurance industry, respectively (“Pfizer Inc,” 2021). With regard to the key success factors, one can note a high efficacy of Pfizer’s product, sophisticated logistics that allow the vaccine to be delivered quickly, as well as its availability.
Target Markets
Based on the demographics of target markets, there is no significant division for vaccine supply. By the end of 2020, “approximately 42% of global participants and 30% of U.S. participants” with diverse ethnic backgrounds had been involved, and half of them were 56-85 years old (“Pfizer and BioNTech,” 2020, para. 8). The key characteristics to take into account include the age of the target participants, their health indicators, chronic lung disease, and contact with COVID-19 patients. In terms of preferences, the target market demands an opportunity to protect from the coronavirus, move around the country and abroad, and participate in the creation of herd immunity. As the vaccine moves on to the market, more and more people want to be vaccinated due to its “100% efficacy and robust antibody responses” (Pfizer, n.d., para. 10). Therefore, the target markets respond to Pfizer’s marketing decisions proactively and quickly.
Market Analysis
SWOT Analysis
Conducting a SWOT analysis can help identify external and internal factors that influence Pfizer’s operations. Regarding the strengths of the corporation, one can highlight its market leadership, continuous technological development, and stable response for any market fluctuations. At the same time, Pfizer’s weaknesses include recurrent supply problems, legal challenges associated with the vaccine’s entry into the global market, and its potential side effects that may be critical.
Pfizer’s opportunities include the global endorsement of the vaccine manufactured, the partnership with BioNTech, another market player, and the effectiveness of clinical solutions to improve product quality. However, there are several threats, in particular, patent infringement, failures to address the current demand, and potential competition in target markets. These factors are crucial to take into account to ensure the corporation’s sustainable operation in the existing market conditions.
Competitor Assessment
One of Pfizer’s competitors in the production of the COVID-19 vaccine is Moderna, the American biotechnological corporation. According to Bottaro (2020), the company uses the same advanced technology as Pfizer, has a solid financial background, and is the first organization to conduct phase 3 clinical trials. However, as Meo et al. (2021) note, Moderna’s vaccine is prescribed to people who are slightly older compared to that of Pfizer, its side effects are more severe, and the market price is higher. These weaknesses are the reasons that hold back Moderna’s leadership position.
Another competitor to Pfizer’s vaccine is Sputnik V manufactured by the Russian Gamaleya Research Institute. Among its advantages, Schraer (2021) highlights the possibility of transportation at a lower temperature, a minimum of side effects, and rapid global distribution. At the same time, according to the author, its efficacy is 92% compared to that 95% -100% of Pfizer (Schraer, 2021, para. 1). Moreover, it entered the market too fast, which raised concerns about the vaccine’s clinical safety, and it cannot enter the American market due to the current sanctions (Schraer, 2021). Therefore, Pfizer management should consider its competitors’ activities to promote the company’s vaccine successfully.
Market Position
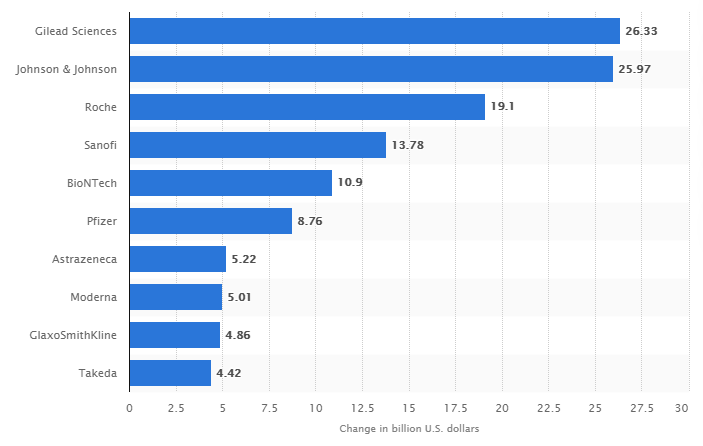
At the moment, Pfizer has a strong market position and is one of the leaders in its industry. However, while comparing the market capitalization of companies involved in the COVID-19 campaign in 2020, the corporation was inferior to a number of American and foreign organizations, as reflected in Figure 1 (“Pharma companies,” 2020). By today, Pfizer has managed to achieve higher profitability in its segment. According to current data, its net margin is 33.51%, and for the first quarter of 2021, the corporation’s net profit increased by 43.28% compared to the same period last year (“Pfizer Inc’s competitiveness,” 2021). All this allows concluding that the company is developing at a fast pace, and its marketing program is successful.
Conclusion
The promotion of Pfizer and its COVID-19 vaccine is successful, particularly compared to last year. The current market indicators prove that the company has earned a positive reputation and become one of the leaders in its segment, despite its competitors’ activities. The analysis of its developmental factors and the target market as a whole allows asserting an effective approach to promoting its main product and maintaining relevant marketing principles.
References
Bottaro, G. (2020). Which Covid vaccine candidates are competing against Pfizer? Proactive. Web.
Centers for Disease Control and Prevention. (2021, June 3). Demographic characteristics of people receiving COVID-19 vaccinations in the United States. Web.
Covid: FDA approves Pfizer vaccine for emergency use in the US. (2020). BBC. Web.
Meo, S. A., Bukhari, I. A., Akram, J., Meo, A. S., & Klonoff, D. C. (2021). COVID-19 vaccines: Comparison of biological, pharmacological characteristics and adverse effects of Pfizer/BioNTech and Moderna Vaccines. European Review for Medical Pharmacological Sciences, 25(3), 1663-1669. Web.
Pfizer (n.d.). All COVID-19 updates. Web.
Pfizer and BioNTech conclude phase 3 study of COVID-19 vaccine candidate, meeting all primary efficacy endpoints. (2020). Web.
Pfizer Inc. (2021). CSI Market. Web.
Pfizer Inc’s competitiveness. (2021). CSI Market. Web.
Pharma companies involved in COVID-19 R&D with the largest market capitalization change, 2020. Web.
Sagonowsky, E. First-to-market Pfizer expects a whopping $15B from its COVID-19 shot in 2021. Fierce Pharma. Web.
Schraer, R. (2021). Russia’s Sputnik V vaccine has 92% efficacy in trial. BBC. Web.
